Precision
RNA Therapeutics
Sovargen was founded on the discovery that somatic mutations arising in the brain may represent a fundamental cause of many intractable neurological disorders. These mutations can occur in neural stem cells and gradually expand over time, contributing to conditions such as focal epilepsy, neurodegenerative diseases including Alzheimer's disease and ALS, and primary brain tumors. We have validated this scientific concept through comprehensive genetic analyses of patient brain tissues and by developing disease models that closely replicate patient-specific genetic alterations.
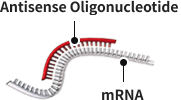
Our core therapeutic platform is based on antisense oligonucleotides (ASOs)—precision RNA medicines designed to selectively bind and regulate disease-causing genes at the RNA level. By targeting the root genetic drivers rather than downstream proteins, ASOs offer high specificity, improved safety potential, and broad applicability across genetically defined brain disorders. This sequence-based approach also enables efficient and scalable therapeutic development.
To address the major challenge of drug delivery to the brain, we utilize clinically established intrathecal administration, allowing our therapies to bypass the blood–brain barrier and reach the central nervous system directly. Through this integrated scientific strategy, Sovargen aims to deliver genetically precise, disease-modifying treatments that address the underlying causes of serious neurological diseases.
- Somatic Variations Genomics
-
Deciphering Mechanisms and Identifying Novel Targets in
Somatic Mutation-Driven Intractable Brain Diseases
- Gene targeted Therapies
-
Development of First-in-Class RNA Therapeutics
Targeting Pathogenic Genes.
Our core platform :
Sovarna

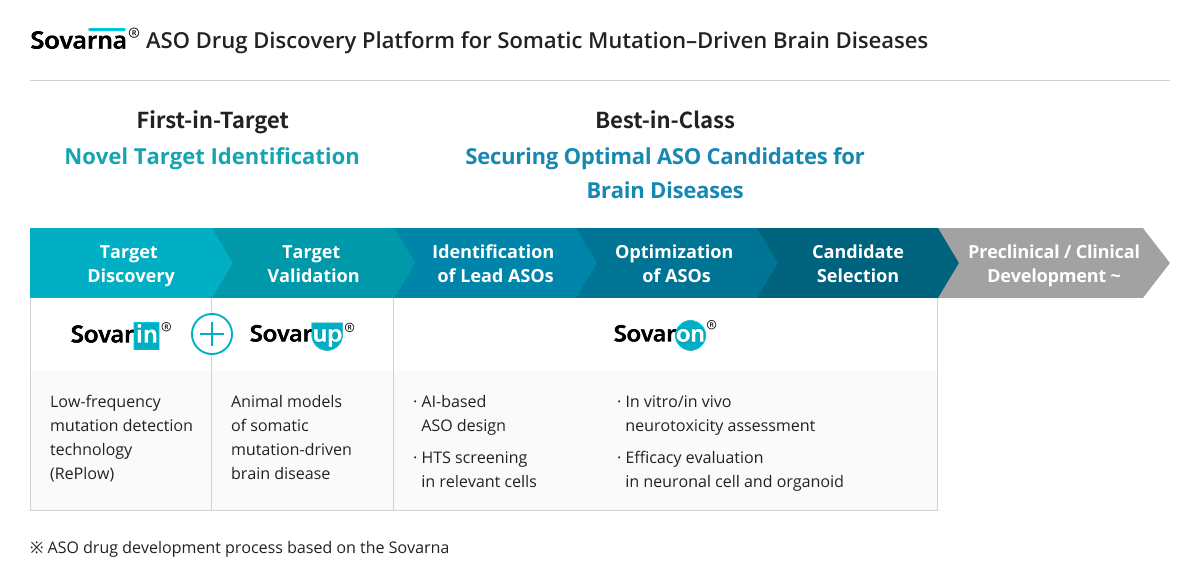
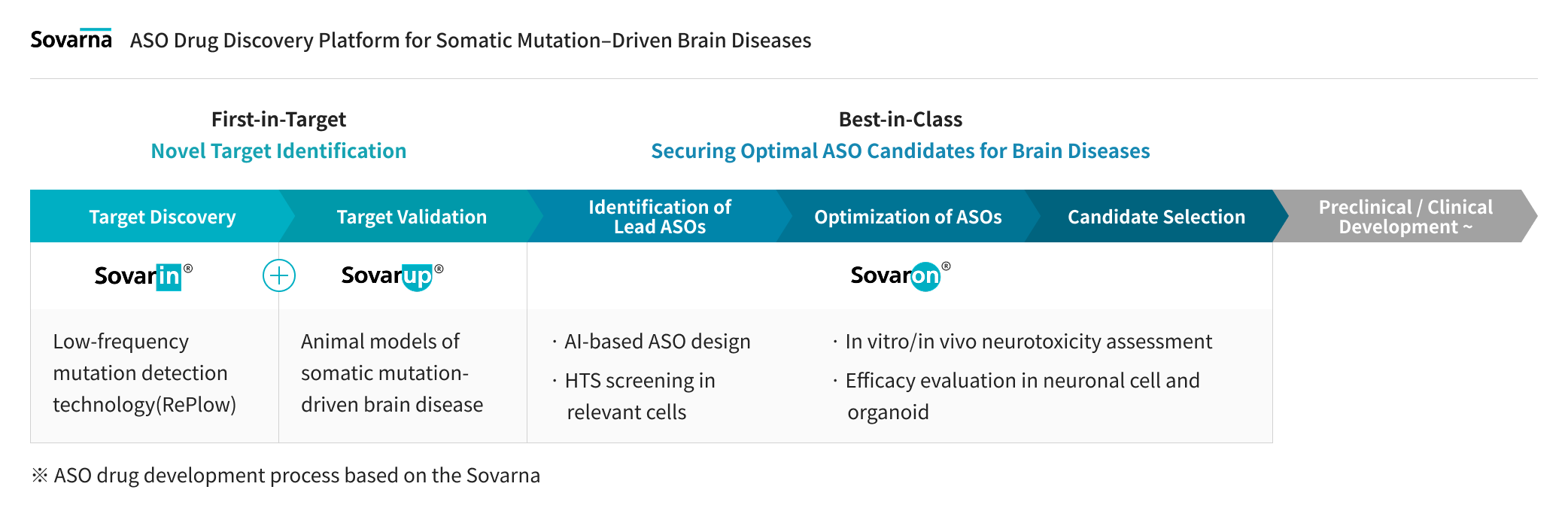
Sovagen has established Sovarna,
a specialized ASO (Antisense Oligonucleotide) drug discovery platform for
intractable brain diseases caused by somatic mutations.
Sovarna is strategically designed to address the three pillars of success in CNS drug development:
Right Target (novel target discovery and validation), Right Tissue (tissue-specific delivery efficacy and safety), and Right Translation (translational research-based drug evaluation).
1.
 Sovarin
Ultra-low level somatic mutation
Sovarin
Ultra-low level somatic mutation
detection technology
Sovarin is a cutting-edge technology designed for the precision detection of ultra-low frequency somatic mutations—a known driver of intractable brain disorders.
Through strategic collaborations with world-renowned hospitals, research institutes, and brain banks, Sovagen has built an extensive biorepository featuring over 3,000 patient brain tissue samples, integrated with comprehensive clinical records.
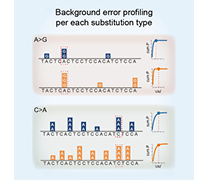
While various bioinformatics methods are currently used for variant interpretation, they often struggle to detect somatic mutations at extremely low levels. To overcome these limitations, Sovagen developed RePlow, a proprietary technology capable of accurately identifying mutations even at a Variant Allele Frequency (VAF) of less than 1%.
- Detection of Brain Somatic Mutations
-
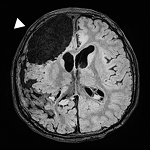
Affected
brain tissue

Matched
unaffected tissue -


Deep sequencing
(e.g. deep WES>500X)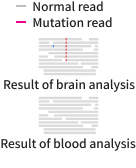
-



RePlow
- Detection of Brain Somatic Mutations
-


Affected
brain tissue

Matched
unaffected tissue
-


Deep sequencing (e.g. deep WES>500X)


-




RePlow
2.
 Sovarup
Brain disease-specific translational research model
Sovarup
Brain disease-specific translational research model
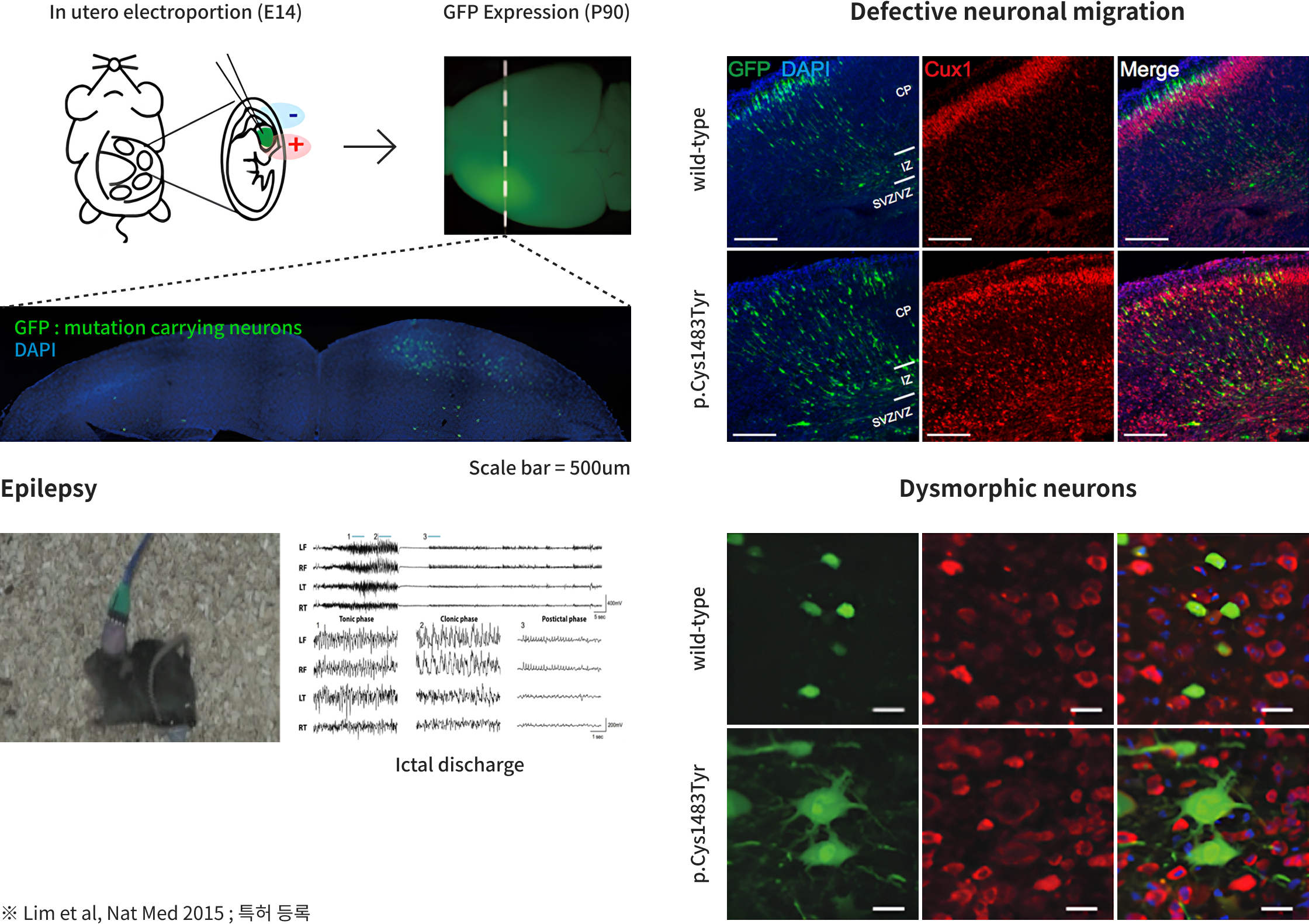
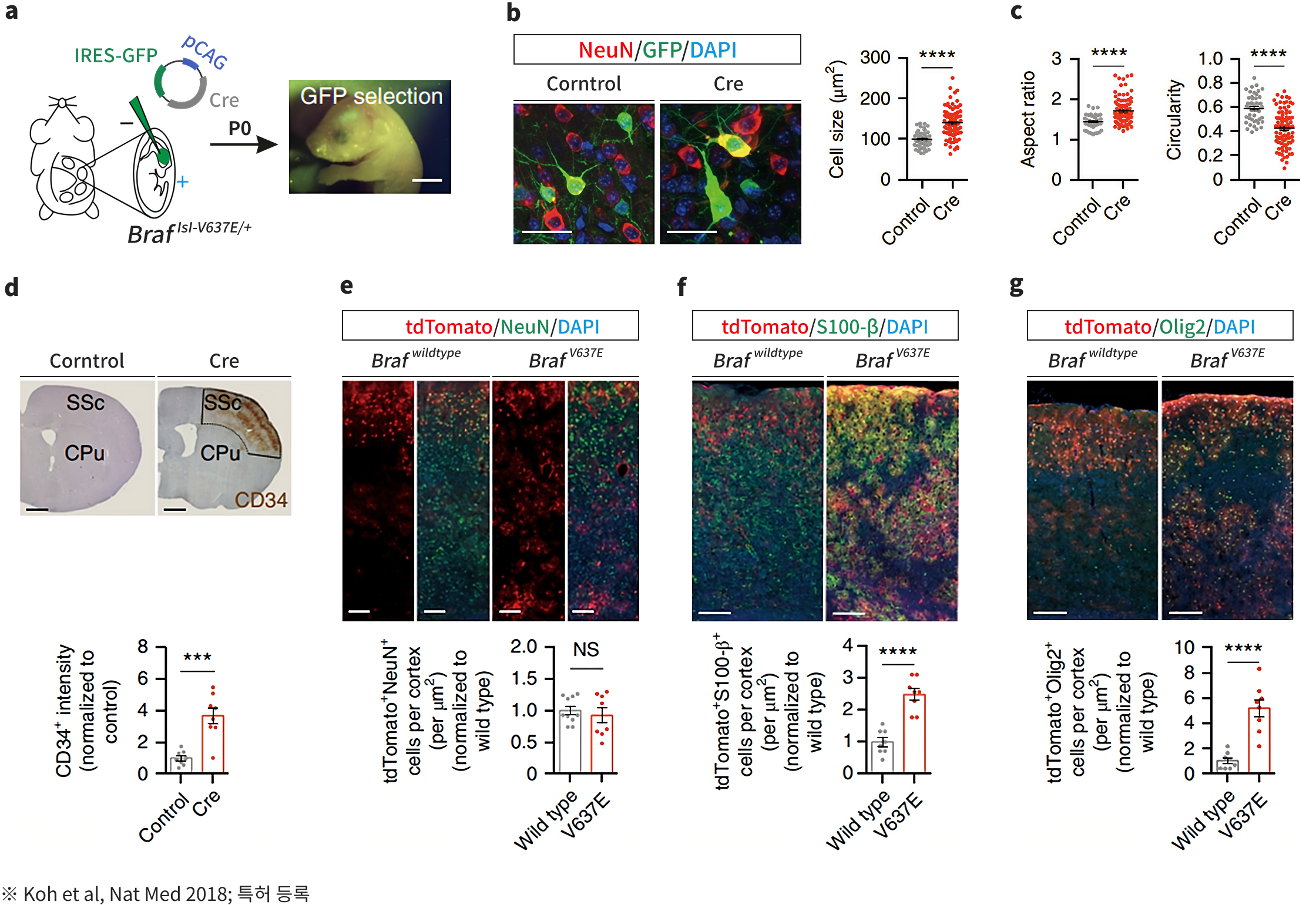
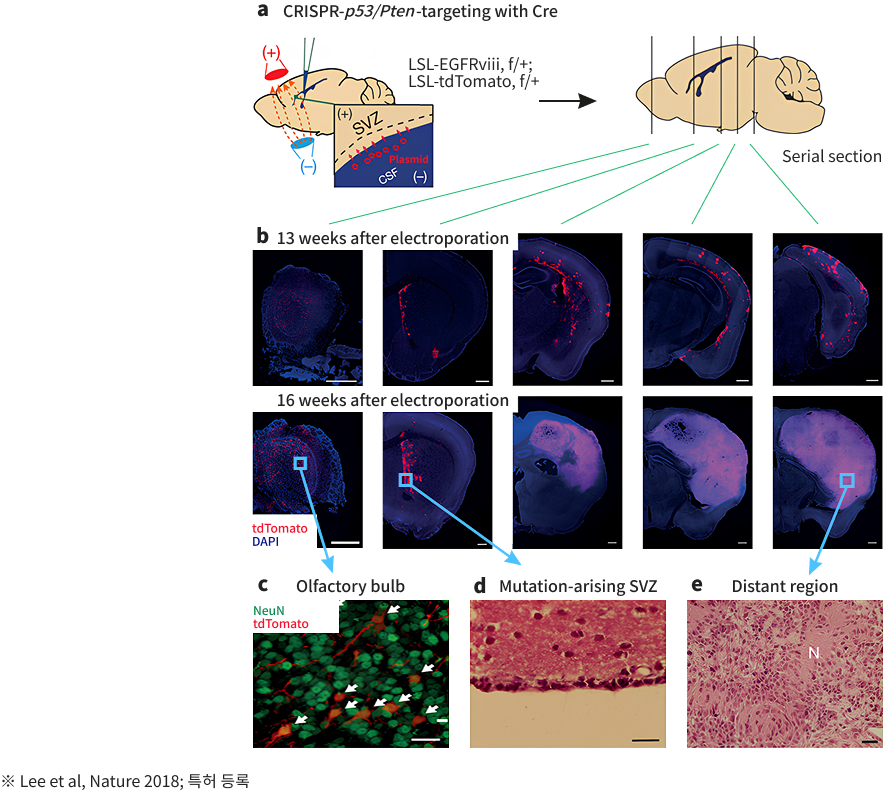
Sovarup is a specialized translational research model designed to revolutionize the study of brain disorders. Unlike traditional models that focus solely on symptoms (phenotypes), Sovarup introduces disease-causing somatic mutations directly into the same cell types where the disease originates in humans. This allows for the precise recapitulation of the entire pathological journey—from the initial onset to the full progression of the brain disorder. By overcoming the inherent limitations of conventional animal models, Sovarup provides high-fidelity translational data optimized to increase clinical success rates. Newly identified targets based on somatic mutations, discovered via Sovarin and Sovarup, represent novel therapeutic opportunities. These insights enable the development of first-in-class innovative drugs and unlock the potential of low-frequency somatic mutations as powerful molecular diagnostic markers.
- FCD II Epilepsy Animal Model
-
- LGG Animal Model
-
- GBM Animal Model
-
3.
 Sovaron
Specialized ASO drug discovery platform optimized for
Sovaron
Specialized ASO drug discovery platform optimized for
neurological disorders
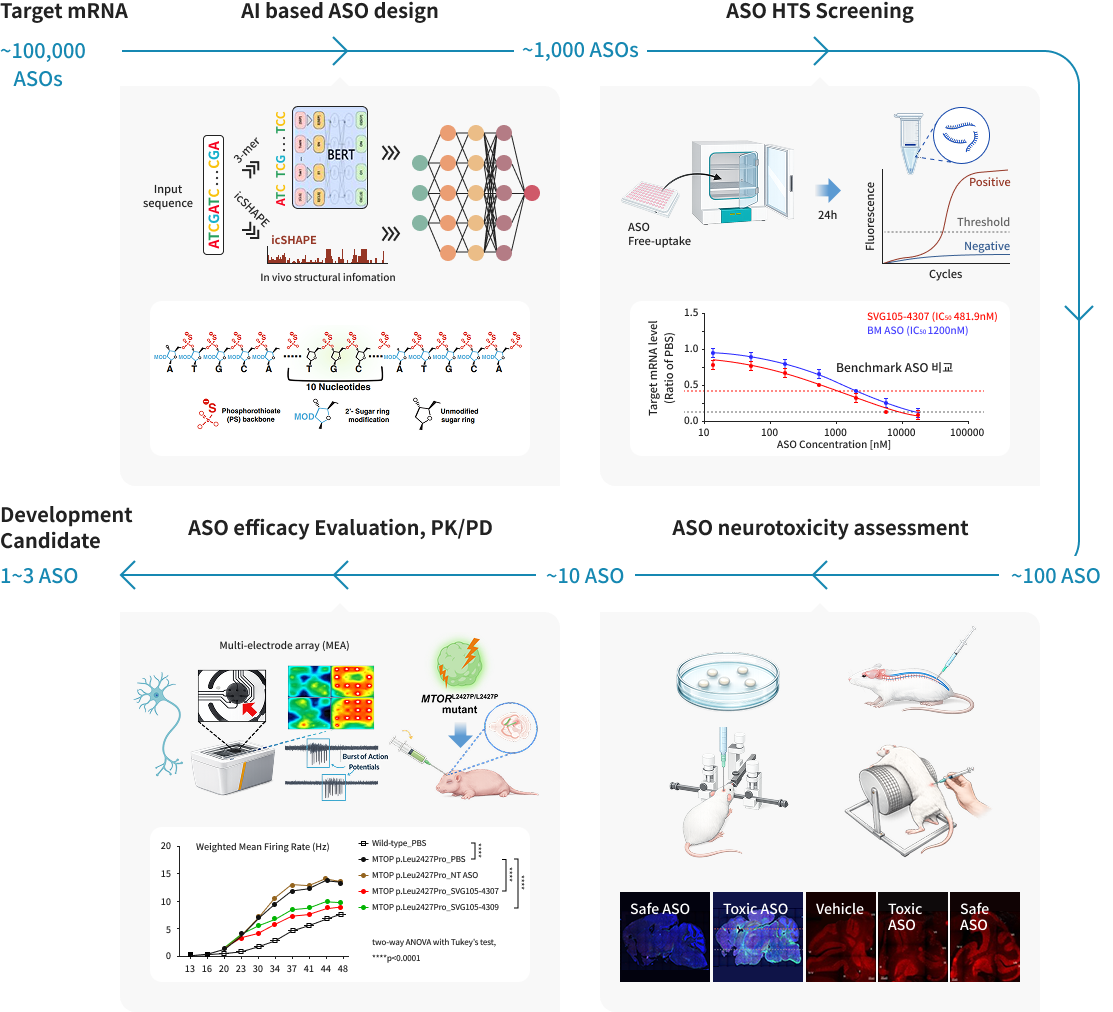
Sovaron is an integrated platform designed to efficiently discover precision Antisense Oligonucleotide (ASO) drug candidates for neurological disorders.
The platform is built upon four core technologies:
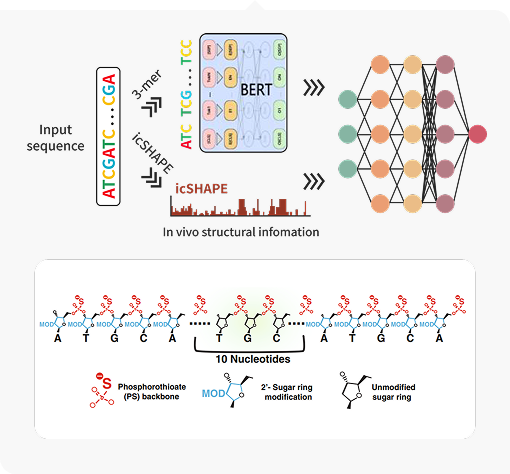
· AI-Driven Design: Utilizing AI to screen vast numbers of ASO sequences and identify candidates with high predicted efficacy and safety,
narrowing down the pool for experimental validation.
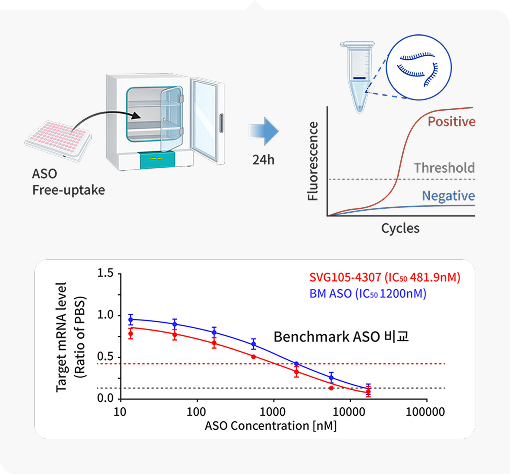
· HTS-Based Screening: A high-throughput screening (HTS) technology that evaluates target mRNA degradation across various cells via
free uptake. This allows for the rapid mass-assessment of initial candidates in a short period.
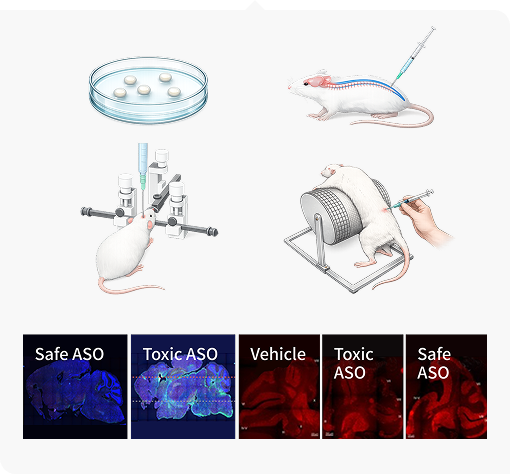
· Neurotoxicity Assessment: Specialized toxicity evaluation using neuronal cells and animal models to early-exclude ASOs
that may trigger acute neurotoxicity, neuroinflammation, or neurodegeneration.
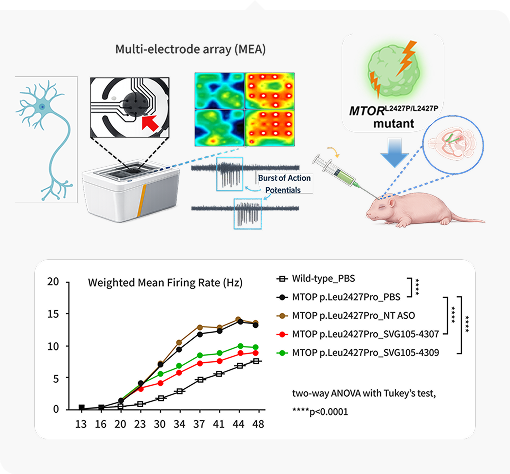
· Translational Efficacy Validation: Verification of therapeutic potential using disease-specific mutant neuronal cells and
organoid models to confirm clinical viability.
Through the systematic application of these technologies and an iterative feedback process, we finalize ASO drug candidates
that ensure both superior efficacy and robust safety.
-
Sovaron-Based ASO Drug
Discovery Process -
-
Target mRNA
~100,000 ASOs
AI based ASO design
~1,000 ASOs
ASO HTS Screening
~100 ASOs
ASO neurotoxicity assessment
~10 ASOs
ASO efficacy Evaluation, PK/PD
1~3 ASO
Development Candidate
Publications
2026
2025

Precancerous Cells Initiate Glioblastoma Evolution and Contribute to Intratumoral Heterogeneity.
Cancer Discov.2025 Jul 3;15(7):1377-1391.

Distant origin of glioblastoma recurrence: neural stem cells in the subventricular zone serve as a source of tumor reconstruction after primary resection.
Mol Cancer.2025 Mar 4;24(1):64.

Efficacy and safety of everolimus for patients with focal cortical dysplasia type 2.
Epilepsia Open.2025 Feb;10(1):243-257.

Associations between physical activities and self-harm behaviour in depression across the genotype: findings from the UK biobank.
BJPsych Open.2025 Jan 31;11(1):e27.
2024
Somatic variant analysis of resected brain tissue in epilepsy surgery patients.
Epilepsia.2024 Dec;65(12):e209-e215.

Whole genome sequencing analysis identifies sex differences of familial pattern contributing to phenotypic diversity in autism.
Gonome Med.2024 Sep 27;16(1):114.

Threshold of somatic mosaicism leading to brain dysfunction with focal epilepsy.
Brain.2024 Sep 3;147(9):2983-2990.

Low-level brain somatic mutations in exonic regions are collectively implicated in autism with germline mutations in autism risk genes.
Exp Mol Med.2024 Aug;56(8):1750-1762.
2023

Ultra-Low Level Somatic Mutations and Structural Variations in Focal Cortical Dysplasia Type II.
Ann Neurol.2023 Jan 26; 93(6):1082-1093.

An agonistic anti-Tie2 antibody suppresses the normal-to-tumor vascular transition in the glioblastoma invasion zone.
Exp Mol Med.2023;55(2):470-484.

Single Cell Analysis of Human Thyroid Reveals the Transcriptional Signatures of Aging.
Endocrinology.2023 Feb 15;164(4):bqad029.

Clinical Features, Neuropathology, and Surgical Outcome in Patients With Refractory Epilepsy and Brain Somatic Variants in the SLC35A2 Gene.
Neurology.2023 Jan 31;100(5):e528-e542.
2022

Analysis of low-level somatic mosaicism reveals stage and tissue-specific mutational features in human development.
PLoS Genet.2022 Sep.

Identification of a pleiotropic effect of ADIPOQ on cardiac dysfunction and Alzheimer’s disease based on genetic evidence and health care records.
Transl Psychiatry.2022 Sep 16;12(1):389.

Protocol to analyze antitumor immunity of orthotopic injection and spontaneous murine high-grade glioma models using flow cytometry and single-cell RNA sequencing
STAR Protoc.2022 Aug 8;3(3):101607.
The ILAE consensus classification of focal cortical dysplasia: An update proposed by an ad hoc task force of the ILAE diagnostic methods commission.
Epilepsia.2022 Aug;63(8):1899-1919.

Epilepsy with SLC35A2 Brain Somatic Mutations in Mild Malformation of Cortical Development with Oligodendroglial Hyperplasia in Epilepsy (MOGHE).
Ann Child Neurol.2022 July;30(3):88-94.

Non-coding de novo mutations in chromatin interactions are implicated in autism spectrum disorder.
Mol Psychiatry.2022 July 15; 27(11):4680-4694.

Brain somatic mutations as RNA therapeutic targets in neurological disorders.
Ann NY Acad Sci.2022 May; 1514(1):11-20.

Efficacy of the Ketogenic Diet for Pediatric Epilepsy According to the Presence of Detectable Somatic mTOR Pathway Mutations in the Brain.
J Clin Neurol.2022 Jan;18(1):71-78.
2021

Detection of Brain Somatic Mutations in Cerebrospinal Fluid from Refractory Epilepsy Patients.
Ann Neurol.2021 Jun; 89(6):1248-1252.

Low-level brain somatic mutations are implicated in schizophrenia.
Biol Psychiatry.2021 Feb 4; 90(1):35-46.

Genetic Architectures and Cell-of-Origin in Glioblastoma.
Front Oncol.2021 Jan 21; 10:615400.

Frequent SLC35A2 brain mosaicism in mild malformation of cortical development with oligodendroglial hyperplasia in epilepsy (MOGHE).
Acta Neuropathol Commun.2021 Jan 6;9(1):3.
2020

Detailed analysis of phenotypes and genotypes in megalencephaly-capillary malformation- polymicrogyria syndrome caused by somatic mosaicism of PIK3CA mutations.
Orphanet Rare Dis.2020 Aug 10; 15(1):205.

SHP2 mutations induce precocious gliogenesis of Noonan syndrome-derived iPSCs during neural development in vitro.
Stem Cell Res Ther.2020 June 3;11(1):209.

SWATH-MS analysis of cerebrospinal fluid to generate a robust battery of biomarkers for Alzheimer’s disease
Sci Rep.2020 May 4;10(1):7423.

Glioblastoma Cellular Origin and the Firework Pattern of Cancer Genesis from the Subventricular Zone.
J Korean Neurosurg Soc.2020 Jan;63(1):26-33.
2019

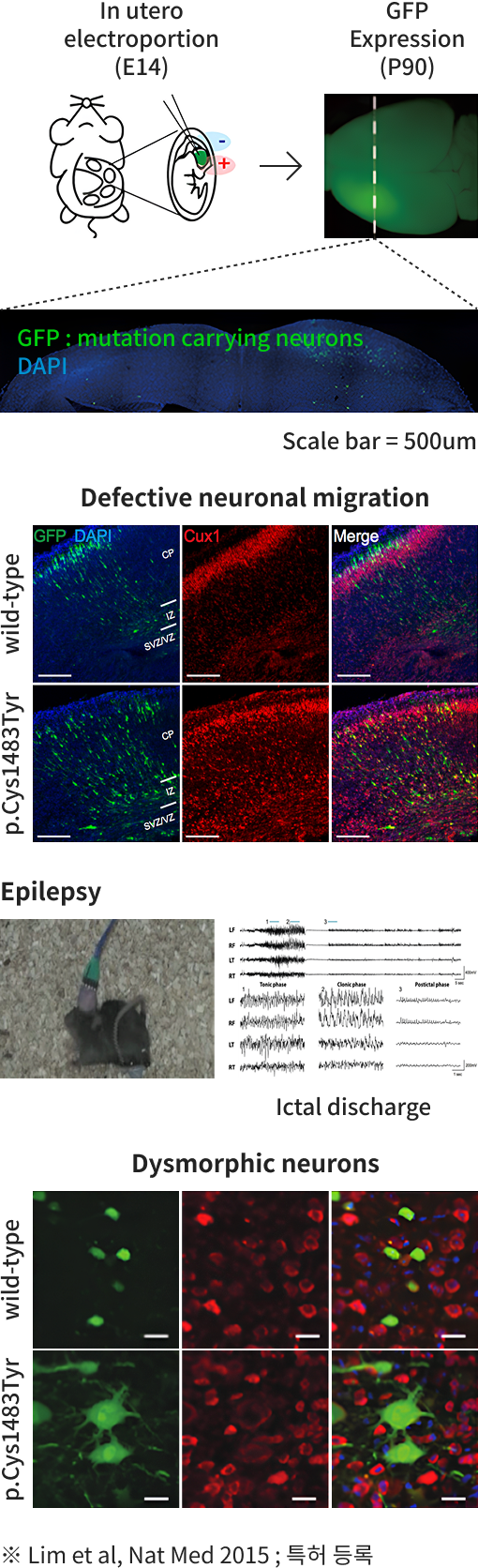
Brain Somatic Mutations in MTOR Reveal Translational Dysregulations Underlying Intractable Focal Epilepsy.
J Clin Invest.2019 Oct 1;129(10):4207-4223.

Precise detection of low-level somatic mutation in resected epilepsy brain tissue.
Acta Neuropathol.2019 Aug 3;138:901-912.

Global analysis of intercellular homeodomain protein transfer.
Cell Rep.2019 July 16; 28(3):712-722.e3.

Brain somatic mutations observed in Alzheimer’s disease associated with aging and dysregulation of tau phosphorylation.
Nat Commun.2019 July 12; 10(1):3090.

Roles of primary cilia in the developing brain.
Front Cell Neurosci.2019 May 14; 13:218.

Mechanistic target of rapamycin pathway in epileptic disorders.
J Korean Neurosurg Soc.2019 May 1; 62(3):272-287.

Extraciliary roles of the ciliopathy protein JBTS17 in mitosis and neurogenesis.
Ann Neurol.2019 April 20; 86(1):99-115.

The use of technical replication for detection of low-level somatic mutations in next-generation sequencing.
Nat Commun.2019 Mar 5; 10(1):1047.
2018

Brain somatic mutations in SLC35A2 cause intractable epilepsy with aberrant N-glycosylation.
Neurol Genet.2018 Dec 5; 4(6):e294.

Miniature ultrasound ring array transducers for transcranial ultrasound neuromodulation of freely-moving small animals.
Brain Stimul.2018 Nov 17; 12(2):251-255.

Brain somatic mutations in epileptic disorders.
Mol Cells.2018 Oct 31; 41(10):881-888.

The origin-of-cell harboring cancer-driving mutations in human glioblastoma.
BMB Rep.2018 Oct; 51(10):481-483.

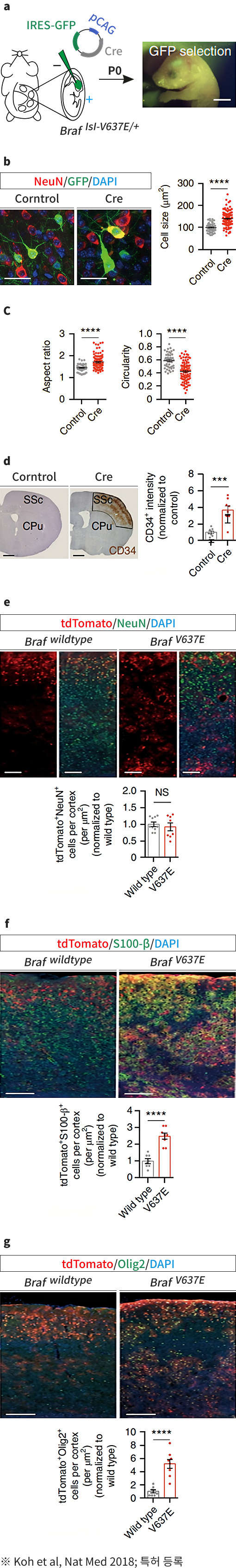
BRAF somatic mutation contributes to intrinsic epileptogenicity in pediatric brain tumors.
Nature Medicine.2018 September 17; 24(11):1662-1668.

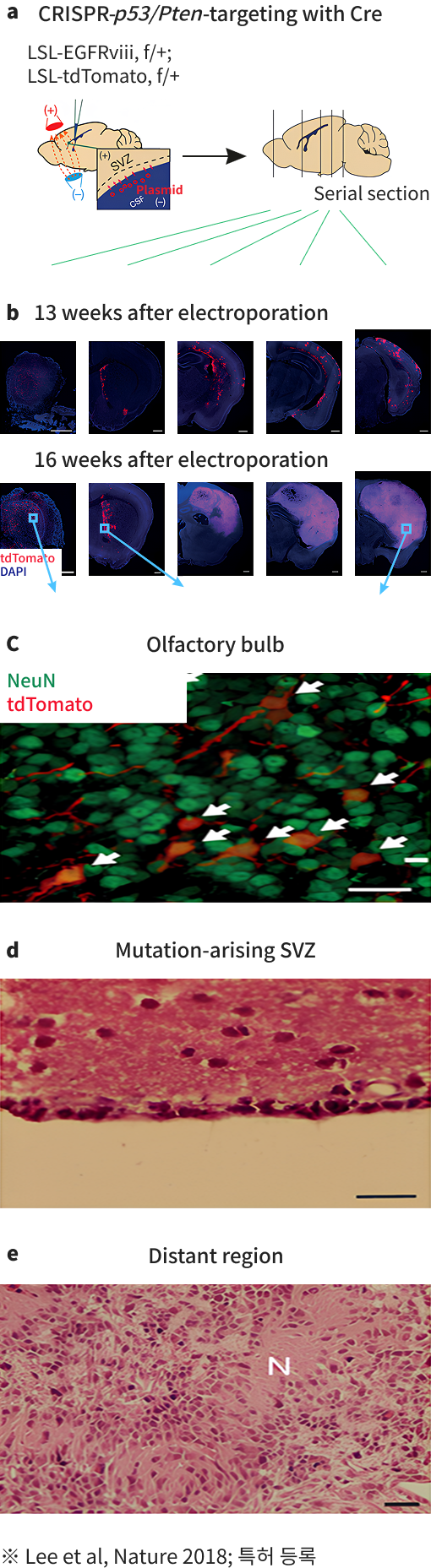
Human glioblastoma arises from subventricular zone cells with low-level driver mutations.
Nature.2018 August 01; 560(7717):243-247.

Brain somatic mutations in MTOR disrupt neuronal ciliogenesis leading to focal cortical dyslamination.
Neuron.2018 July 11; 99(1):83-97.e7.
2017
2015
2012